Specialized Blood and Tissue Processing for Advanced Pharmaceutical Manufacturing
With over 36 years of industry experience, Vista Biologicals delivers precision-engineered blood and tissue processing as part of our comprehensive pharmaceutical services. From isolating mononuclear cells (MNCs) from cord blood to freezing umbilical cord tissue for long-term storage, our processes support advanced research applications and clinical-stage development.
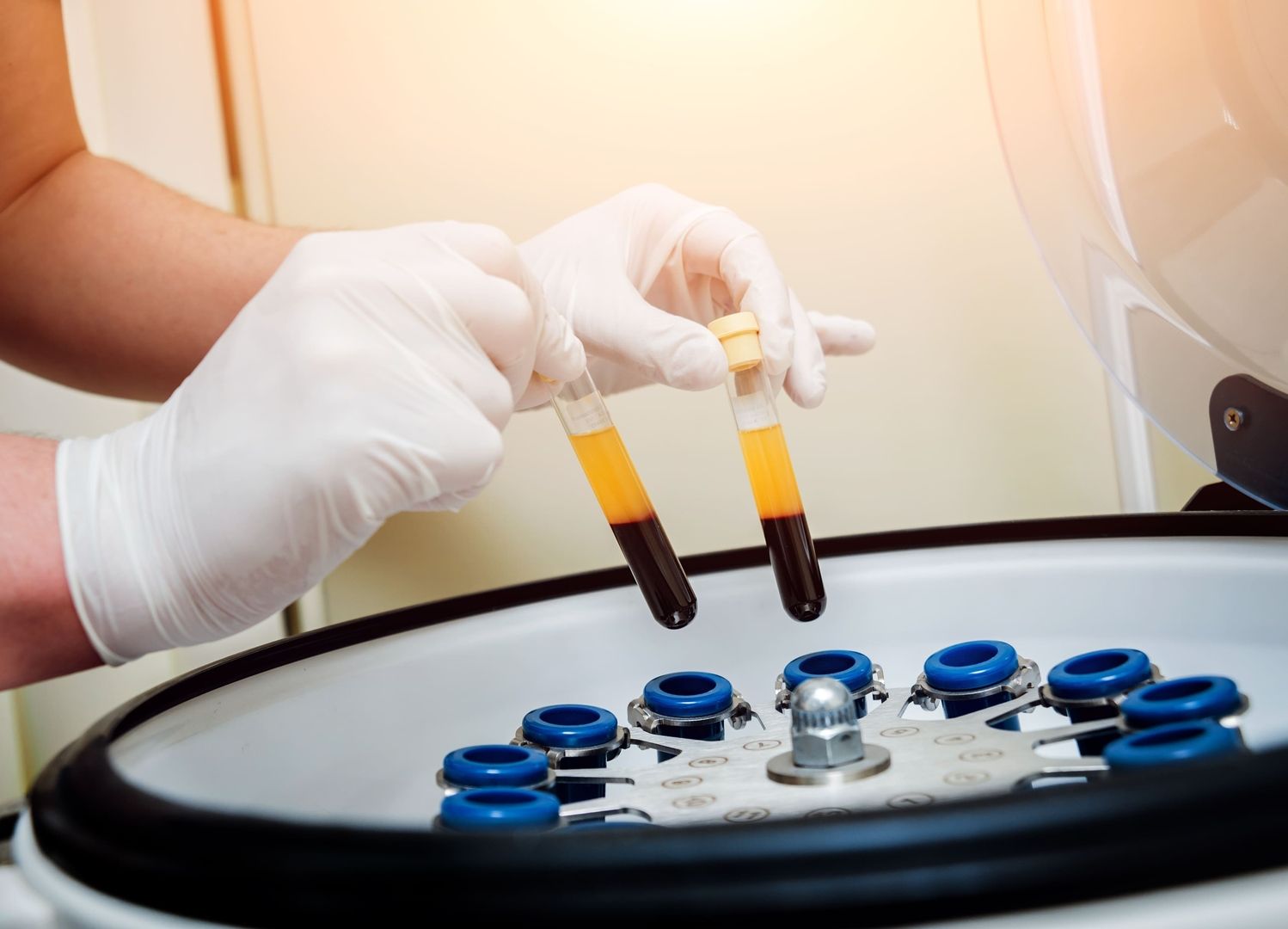

Blood and Tissue ProcessingStem Cell Research
Our capabilities extend into the realm of pharmaceutical manufacturing. We prepare cells, plasma, and tissue-derived fractions under GMP-informed conditions, supporting your pipeline from early-stage exploration to regulated supply. We also collaborate on unique niche applications – for example, processing blood and plasma to generate adsorbents used in antisera and toxin-neutralization workflows.
Key Capabilities
- Mononuclear Cell Isolation and Cell Banking
We isolate MNCs from cord blood and store them under validated cryogenic conditions for downstream use in biologics or investigational cell therapies. - Tissue Processing and Storage
Umbilical cord tissue is processed for epithelial and mesenchymal stem cell sources, offering a foundation for grafts, corneal resurfacing, dermatologic repair, diabetic wound therapies, and more. - Blood and Plasma Processing for Downstream Applications
Our facility handles whole-blood and plasma processing to supply adsorbents for antisera manufacture, snake-venom neutralization, and other specialized toxin-management projects.
Why Choose Vista Biologicals
When you engage our team, you are partnering with experts whose core competencies span both pharmaceutical services and pharmaceutical manufacturing. We bring:
- a track record of more than three decades in cell culture, tissue processing, and biologics support.
- an infrastructure aligned with GMP principles, enabling a seamless transition from development into regulated manufacturing.
- customizable workflows that adapt to your project’s specific needs – from niche stem cell sourcing to broader process scale-up for clinical supply.
Whether your priority is pioneering stem cell modalities, novel tissue-derived therapeutics, or fine-tuned blood and plasma fractionation, Vista Biologicals stands ready to support your program with disciplined operations, rigorous quality systems, and collaborative responsiveness.
